Earlier the subject of powders was directed from a physicochemical standpoint. The unit processes involved in incorporating powders in solid dosage forms must also be considered. Pharmaceutical Solid dosage forms can be divided into tablets , capsules, granules for oral delivery, and inhalation products. Note that solid particulates might also play a role in certain parenterals in the form of reconstituting products.
Solid dosage forms are the most desirable final products of a development process that begins with drug discovery and proceeds through bulk product manufacturing, preformulation, and formulation characterization to one of the products mentioned.
illustrates an abbreviated sequence of steps through which the drug passes to the final dosage form.
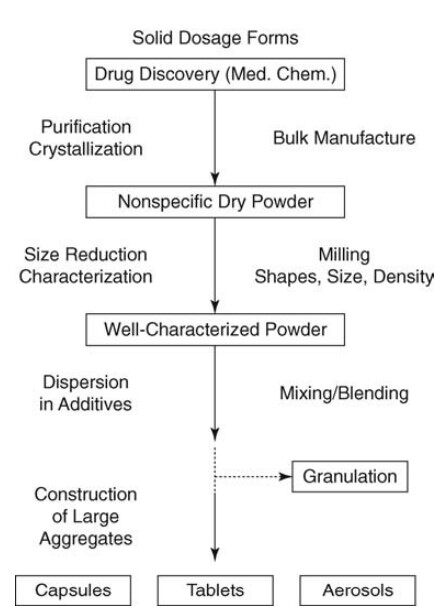
Most solid dosage forms are intended for oral ingestion. The drug released from the dosage form is available at the site of absorption or action within the gastrointestinal tract. Preformulation studies are required before a formulation is developed. By studying the properties of the drug, it is possible to delineate a course of action for composing the formulation. The properties studied are organoleptic properties, purity, particle size, shape, and surface area, solubility, dissolution, parameters affecting absorption (dissociation constant, partition coefficient), crystal properties and polymorphism, stability (chemical and physical), compatibility (with excipients and potential packaging materials), and miscellaneous physicochemical properties like density, hygroscopicity, flowability, compressibility, and wettability.